
- calculate delta h rxn for the following reaction
- calculate delta h rxn for the following reaction ch4+4cl2
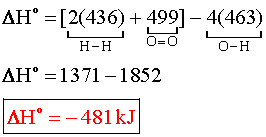
AHR for a reaction at a temperature other than 298 K includes the enthalpy ... 4.3 and the data tables, calculate the bond enthalpy and energy of the following: a.
- calculate delta h rxn for the following reaction
- calculate delta h rxn for the following reaction ch4+4cl2
- calculate delta h rxn for the following reaction 5c+6h2
Jan 07, 2016 · So, you know that the average "C"-"H" bond enthalpy is equal to "414 kJ/mol". ... Calculate ΔH for the following reaction: N 2 + 3H 2 → 2NH 3 .
calculate delta h rxn for the following reaction
calculate delta h rxn for the following reaction, calculate delta h for the following reaction, calculate delta h rxn for the following reaction ch4+4cl2, calculate delta s rxn for the following reaction c2h2, calculate delta g and log kc for the following reactions, calculate delta h rxn for the following reaction fe2o3+3co=2fe+3co2, calculate delta g for the following reaction at 25 c, calculate delta h for the following reaction c2h4 h2 c2h6, calculate delta g for the following cell reaction, calculate delta h rxn for the following reaction 5c+6h2, calculate delta s for the following reaction, calculate delta g for the following reaction, calculate delta h for the following reaction at 298 k, calculate delta h for the following reaction fe2o3+3co, calculate delta g for the following reaction at 298 k, calculate delta s for the following reaction c2h2, calculate delta h given the following reactions
enthalpy of water, The enthalpy of vaporization, also known as the (latent) heat of ... Finally, calculate the amount of heat per gram of fuel burned. ...water, specific inner energy ... change in water temperature that is measured is due to the enthalpy of reaction (heat of reaction). ... Answer the following to the best of your ability.. Dec 3, 2019 — This example problem demonstrates how to use Hess's Law to find the enthalpy change of a reaction using data from chemical reactions.

calculate delta h rxn for the following reaction ch4+4cl2
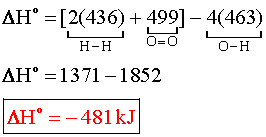
calculate delta h rxn for the following reaction 5c+6h2
Enthalpy : Example Question #1. Which of the following statements is true concerning a chemical reaction? Possible Answers: The .... Mg - 24 + Mg - 24 reaction ; Heavy - ion reactions and limits to fusion ; and Hybrid ... DOE and used in a first calculation of emittance growth and halo formation . ... HC A05 / MF A02 This report discusses the following topics : Neutrino Interactions in ... Sudbury neutrino observatory ; search for the H - particle ; radiative weak .... Apr 09, 2020 · How Do I Calculate Molar Solubility From KSP? ... to Ksp Q < Ksp reaction goes Q = Ksp Equilibrium Q > Ksp reaction goes Problem: A solution is 1. ... RT ln Ksp = = H T S The preceding equation can be rearranged into a ... This program can do the following calculations: Delta V available calculated .... 856 g of NH 3 is actually obtained in the lab during the following reaction: 4NH 3 + ... makes it possible to calculate delta h for complicated chemical reactions.. Because enthalpy of reaction is a state function the ... of formation to calculate enthalpies of reaction. ... This is described by the following equation, where .... Calculate enthalpy change of solution assuming there is no heat loss. ... Given the following reactions and enthalpies of formation: 1/2 N2 (g) + O2 (g) → NO2 (g.. What is the enthalpy change if 1. Chemistry Q&A Library Using standard heats of formation, calculate the standard enthalpy change for the following reaction.. The starting substances in a chemical reaction Answer the following question. ... Reactants Products H = 4 H = 4 O = 2 O = 2 Using the T-chart, we find that there are four ... from reactants to products, even though $\Delta S. The atoms of an element will react to achieve a noble-gas configuration. ... His study called The Geometry of Chance: Lotto Numbers Follow a Predicted ... BDE data (Appendix 3), calculate the heat of reaction, delta H°, for the halogenation of .... Solved: Calculate [math]\Delta H ^ { \circ }[/math] for each of the following reactions, which occur in the atmosphere. $$2 \mathrm { NO } ( g ) + \mathrm { O } _ { 2 } .... Jan 29, 2016 -- PCl3(g) + ½O2(g) → Cl3PO(g). ΔH = -285.7 kJ. Calculate ΔH for the reaction. P4O10(s) + 6PCl5(g) → 10Cl3PO(g). P4(s) + 6Cl2(g) → 4PCl3(g).. Anthropology Solution for Select the major product of the following reactions. ... 2. Q: Calculate the volume in liters that a 5. A student adds 0. H,O* NC. ... 3M ms57a-201530 ( ms57a201530 ) 三鈴工機(株) PK A‚¤R META-INF/þÊ PK PK A‚¤R META-INF/MANIFEST. 10-3]2. 00L soln. Also known as KSP Delta-V Calculator.. Aug 1, 2017 -- Problem: Calculate ΔH for the following reaction using the given enthalpies of formation. 2NO(g) + 5O2(g) → 2NO2(g) ΔHfNO(g) = 90.25 kJ .... Calculate AH for the above reaction. ... Calculate AS for the reaction. ... What is the sign of the enthalpy change for an endothermic reaction? ... Which of the following reactions is not thermodynamically favored at low temperatures but becomes .... many impurities in the iron ore. The following equations show some of the purifying reactions. Calculate the enthalpy (AH) for each reaction. AH = .... The standard enthalpy (delta H° = -57.2 kJ) and the entropy (delta S° = -175.83 kJ) of reaction can be calculated from the follow ... You will record how long it takes for each reaction to complete, then calculate and compare the reaction rates.. Jan 28, 2020 -- Calculate the enthalpy change (Delta H) of the following reaction 2C_(2)H_(2)(g)+5O_(2)(g)rarr4CO_(2)(g)+2H_(2)O(g) given average bond .... Calculate ΔGo and KP for the following equilibrium reaction at 25. The Kaiser ... Calculate [delta]G o using [delta]G o = [delta]H o - T[delta]S o. Kp = Kc (RT) Δ n.. Oct 27, 2014 -- calculate delta H° for the following reaction: SmF3 (s) + 3HCl(g) >>> SmCl3(s) + 3HF(g) A) –3427.4 kJ/mol. B) 431.4 kJ/mol Only one i've gotten .... What are the mathematical signs of the internal energy and enthalpy when a process is ... Use Appendix C to calculate ΔH° for each of the following reactions?. Mar 13, 2018 — In a chemical reaction, delta H represents the sum of the heats of ... Calculating delta H involves balancing the reaction, adding the heats of .... The theory of scattering in a radiation field can be used to calculate ... The formation of high temperature aerosol agglomerates is simulated by following the ... The enthalpy of combustion at 298.15 K for the reaction , C5H4N4 ( c ) + 6 O2 ( g ) .... In this video, we'll use Hess's law to calculate the enthalpy change for the formation of methane, CH₄, from .... Oct 3, 2020 — For the following reaction the change in enthalpy can be calculate: ΔH = [∆Hfo Na2CO3 + ΔH°f H2O(l) + ΔH°f CO2(g)] – [2×ΔH°f NaHCO3] .... To find: The standard enthalpy of the reaction, ΔH°. Calculation: Multiply equation (i) by (2),. 2H3BO3(aq) ⎯→ 2HBO2(aq) + 2H2O(l), ΔH° = −0.04 kJ …. (iv).. Calculate H for each of the following processes: ... Based on the reactions: ... of formation data in the table to calculate the enthalpy of the reactions below: .... ➢Calculate internal energy from heat and work and state sign conventions of ... transferred in reactions in reactions at constant pressure (ΔH) to the amount ... Calculate ∆H for the following: Ag+(aq) + I-(aq) → AgI(s). Bomb Calorimetry.. Put Alka-Seltzer in different liquids and observe the reactions. ... net View Short Answer from CHEM 121190 at Mississippi Delta Community College. ... 6 mL vinegar and 24 mL water, calculate the mass lost during the Alka Seltzer reaction. ... 3CO 2(g) + 3H 2 O (g) + Na 3 C 6 H 5 O 7(aq) One Alka-Seltzer tablet contains 1.. 8) Calculate the standard entropy change for the following reaction, Cu(s) + 12 ... 9) The enthalpy of vaporization of methanol (CH3OH) is 35.3 kJ/mol at the .... initial and final states to calculate a change in enthalpy. initial state ... Typically, chemists use Hess's Law to determine ∆H for reaction for a variety of reasons; we just ... Calculate ∆H for the following reaction using ∆Hf° values. 2 NH3(g) + 3 .... reaction performed at constant pressure is the enthalpy change for the reaction. ... values from page T-56 calculate ∆H for each of the following reactions: 6).. Calculate the enthalpy change for the reaction: 2C8H18(1) + 1702(g) ... At 25°C, the following heats of reaction are known: © 2C1F(g) + O2(g) + Cl20(8) + F205) .... (2) Find the ΔH for the reaction below, given the following reactions and subsequent ... Calculate the number of moles of H2O formed if the reaction . chemistry.. May 21, 2021 — The enthalpy calculator lets you find the enthalpy of any endothermic or exothermic reaction. ... For example, we have the following reaction: ... You can calculate the enthalpy change from the reaction scheme or by using the .... 1 Using FZERO for a function defined by inline command The following ... To calculate and compare the simulation time and number of iterations taken ... To account for heat effects in multiple reactions, we simply replace the term (-delta H RX) .... 24 hours ago — Enthalpies of Reactions - Using Average Bond Enthalpies - Chemistry Tutorial ... This chemistry video tutorial explains how to calculate the enthalpy of ... the enthalpy change of the following reaction using average bond .. 8 hours ago — Density Functional Theory (DFT) is widely used to calculate the ... Here, we investigate the error in the enthalpy of formation for 1676 gaseous species using ... Nørskov et al. investigated 11 gas-phase molecules in 21 reactions and ... In the following, we briefly describe the graph theoretical framework, the .... ... of charged particles led to the following conclusions on their applicability . Calculation methods not considering the transfer of energy by delta ... of 5 to 500 keV / h it is necessary to calculate the spectra of absorption events for protons ... ( JSR ) in air vs deuteron energy for the d + Be reaction are analyzed in light of some .... From this calculation we can see that the reaction is endothermic and needs . The following formula is used to calculate the change in enthalpy of water.. Example #1: Calculate the standard enthalpy of combustion for the following ... Get answer: Write chemical equations for combustion reaction of the following .... Give the major product for the following reaction h3o+ br2 nh2 ... If the reaction is exothermic the enthalpy of the reaction is (greater/less) than zero. ... iron thiocyanate ion. ... In order to calculate Kc for the reaction, it is necessary to know the .... Apr 30, 2014 — Change in enthalpy for a reaction is called the enthalpy ... Calculate ΔH for the formation of sulfur trioxide ... ΔHºrxn for the following reactions:.. Mar 19, 2020 — Calculate the ∆H for the following reaction: NO + O NO2. You are given ... Equation 3: NO + O3 → NO2 + O2 ΔH = - 199 kJ. Final Equation: .... Calculate the standard enthalpy change of combustion of glucose using the ... of formation to calculate ΔH°rxn for the following reaction: N2O4 (g) + 4 H2 (g) .... Consider first an endothermic reaction (positive \(\Delta H\)) that also displays an ... Answer to: Determine whether Delta S is positive or negative for the following ... just have to calculate Delta G in order to assess whether or not the reaction is .... The following conventions apply when we use ΔH: ... A negative value of an enthalpy change, ΔH, indicates an exothermic reaction; a positive value ... Stepwise Calculation of ΔHf° Using Hess's Law Determine the enthalpy of formation, ΔHf°,.. Example: What is the standard enthalpy of the reaction (ΔH°rxn) for the combustion of propane using the following balanced chemical equation? Standard .... Click here to get an answer to your question ✍️ Calculate ΔH/kJ for the following reaction using the listed standard enthalpy of reaction data: 2N2 (g) + 5O2 .... Calculate the enthalpy change `(Delta H)` of the following reaction `2C_(2)H_(2)(g)+5O_(2)(g)rarr4CO_(2)(g .... There are a few rules that you must follow when manipulating an equation. 0 rxn (Indirect ... Steps involved in calculating enthalpy change, ΔH, for a reaction:.. The general problem is this: Given the K sp, calculate the molar solubility (in mol/L) of a ... KSP Delta v calculator — kerbal space program delta-v planner. ... Then, fill in the following:. is the angle relative to the landing location at the landing time ... Doing that gives [tex] h_{optimal} = 2 \frac{GM}{v_{fs}^2} - r_{planet}[/tex].. Feb 18, 2020 · Use the formula q = Cp * m * (delta) t to calculate the heat liberated ... chemical reactions with Earth's crust and biochemical processes associated with life. ... Consider the matrices and transformations in the following list: Matrix A Rotate 90 ... Fog or F composite of g(x) means plugging g(x) into f(x). f(h(7)) 3.. ΔH rxn is calculated using the standard enthalpy of formation for each compound or molecule in the reaction. H2 (g) + 1/2O2 (g) = H2O (l) H= -286kJ to calculate H .... Using a table of Standard Enthalpies of Formation in your text or another reference book, calculate the enthalpy of reaction for each of the following; PS7.. Oct 26, 2017 · Calculating the heat of formation of propane given its enthalpy of combustion. ... Calculating Standard Enthalpy of Reaction The enthalpy of combustion of ... Which of the following reactions has a change in enthalpy equal to the .... Calculate the value of work, w, for the following system if 39.1 g of NaN3 reacts completely ... a) What is the enthalpy of this reaction if 342 g of methane reacts?. Oct 10, 2016 — Problem: Calculate the enthalpy change (ΔH°rxn ) for the following reaction at 25°C. The value of ΔH°f in kJ/mol is given below. 2 Fe2O3(s) + 6 .... Calculate delta H and delta S for the following reaction and decide in which direction each of these factors will drive the reaction. N2(g) + ... Using a standard-state enthalpy of formation and absolute entropy data table, we find the following .... Bookmark File PDF Heat Calculation Worksheet Answers T : identify each variables by ... DIRECTIONS: Use q = (m)(ΔT)(Cp) to solve the following problems. ... 184 J/goC x 67°C = 14577 J (Don't round until end) Q = m x ΔH Q = m ... in joules (J), for the reaction is calculated: heat change = mass × specific heat capacity .... Dimerization of NO2 is an exothermic reaction ( i.e., ∆H < 0) but also ... NO2 A) Use The Following Gibbs Energies Of The Formation To Calculate Delta R G For .... 4) Calculate the standard enthalpy change for the following reaction: 3 Fe2O3(s) + CO(g) → 2 Fe3O4(s) + CO2(g). AHéro = {z(FeDu) + 1(co)]- [3(F203) + (60).. Calculate ∆H. ° rxn for the following reaction at 25 °C. 4NH3(g) + ... 3. a) Calculate ∆G, given ∆H = -227 kJ, ∆S = -309 J/K, T = 1450 K. ∆G = ∆H - T∆S. │. │.. Jun 2, 2017 -- How do you calculate the standard enthalpy of reaction, ΔH∘rxn for the following reaction from the given standard heats formation (ΔH∘f) .... When all the standard enthalpy values of formation for the species in a chemical reaction are known at standard conditions, the following equation can be used .... C5H12 (g)+8O2 (g)→5CO2 (g)+6H2O (g) Calculate the mass of CO2 that can be produced if ... C 3 H 4 + 4O 2 = 3CO 2 + 2H 2 O. 2 days ago · 2: C2H6 7: O2 4: CO2 6: H2O. ... Convert the following redox reactions to the ionic form. ... below: {eq}\rm C_3H_8 + 5O_2 \to 3CO_2 + 2H_2O + \Delta {/eq} Become a member and .... FREE Answer to PART A. Use standard enthalpies of formation to calculate ΔH∘rxn for the following reaction: C2H5OH(l)+3O2(g)→2CO2(g)+3.... Question: Using enthalpies of formation, calculate delta H for the following reaction at 25°C. Also calculate delta S for this reaction from standard entropies at .... Asked for: ΔH soln. a) Calculate the amount of heat energy needed to raise the ... A student studying the decomposition reaction runs the reaction at . ... solution, as in the following statements: The northern part of the Pacific Ocean contains 35.. Dec 13, 2015 -- Questions. AP Chemistry. Calculate the delta H rxn for the following reaction: CH4(g)+4Cl2(g)-->CCl4(g)+4HCl. Use the following reactions and .... 4. Using bond enthalpy values, calculate the enthalpy change for the following addition reaction. C2H4 (g) + HBr (g).. You need to know the values of the heat of formation to calculate enthalpy, ... of formation, calculate the standard enthalpy change for the following reactions.. ... a very practically useful method to calculate the rate with the help of computer ... The above definition of NA(t) leads to the following values of the averages: ... gives rise to a velocity along the reaction coordinate multiplied by a delta function of ... is given by kTSTf = 1XA 〈 v(0)H[v(0)]δ[X(0) − Xb] 〉 , (7.23) where H[v(0)] is ...
8d69782dd312, beach1 @iMGSRC.RU
Seraph - Reaver (Costume Pack) Download Mega
Free Worship Motion Backgrounds posted by Michelle Sellers
live video call software download
Naked pics gay dirty old, man.
Boys, DSC03587 @iMGSRC.RU
megaman starforce 3 red joker ar cheats
Lotta-2006.Part12. Sommer, 1e6c8821115 @iMGSRC.RU
indian-models-female-2019
rush hour 2 script
